In September 2017, when I defended my thesis on the biosynthesis of diketopiperazines (never heard of diketopiperazines? Impossible, for sure you have already found in your mailbox an unwanted Cialis commercial!) in Streptomyces (you know, those weird filamentous fungi that are actually bacteria and produce more than 60% of medically relevant antibiotics), I started to search for postdoc offers and rapidly stumbled upon the one from the Simonetta Gribaldo’s lab. The subject was studying the evolutionary transition between the diderm (i.e., Gram-negative) and monoderm (i.e., Gram-positive) cell envelope organization in Bacteria, using Veillonella parvula as a model. I was surprised to find that this bug – a very small (below 500 nm in diameter) anaerobic coccus thriving in our dental plaque – was a diderm Firmicute. At the university I had always been taught that Firmicutes are the low GC gram-positives, and I didn’t know (together with most of my fellow microbiologists) that some of them have an outer membrane. My surprise was even greater when I learned that the very first description of the diderm architecture in 1964 was for shown on an electron micrograph on this precise bug1. Hearing that Simonetta was the nicest, kindest person on the Earth, albeit with a Sicilian temper, made my determination to get this postdoc even stronger, all the more so that I had the surprise to discover that the main collaborator was the guy in charge of the Institut Pasteur Microbiology Course I’d taken during my master degree studies, Christophe Beloin. Fascinated by evolution since I’d first heard about this concept during my childhood, I was like “this offer is for me”!
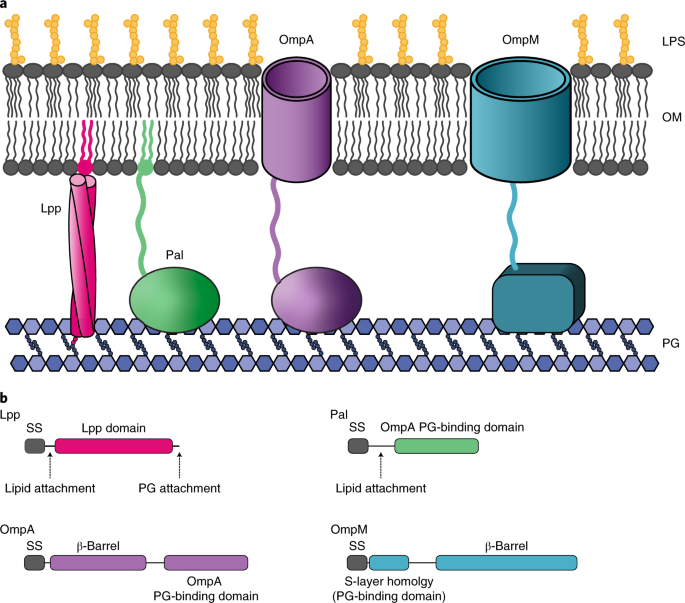
The project involved working on the freshly developed model of V. parvula, for which nascent genetic tools had been established only one year before2, with hope that it would enlighten us how the diderm to monoderm transition happened. Why using a diderm Firmicute? Because in Firmicutes this transition happened independently at least three times3. One of the aims was focusing on how Veillonella tethers its OM to the peptidoglycan, as we thought (and still think) that losing this attachment was one of the major mechanisms involved in the diderm-to-monoderm transition. The plan was to generate the mutant of the neighbouring genes coding for the three OmpM proteins present in Veillonella, analyse the phenotypes of the constructed mutant, and then complement it. It all sounds so simple when you read our paper. Well, it was not.
Nothing but the first step – constructing the mutant – was a challenge. Amplifying anything in the region of the genome containing ompM1-3 stuffed with heavy repetitions was a nightmare. PCR wouldn’t work at all, or would give multiple bands, or amplify not what I wanted… When I managed to obtain my PCR fragments, it was only to find out in the next step that there was no way of making the PCR assembly. The technique I used to make each and every other V. parvula mutant would inexorably fail for ompM1-3. I had to use Gibson assembly and clone our “upstream – resistance marker – downstream” fragment into a vector.
Thankfully the transformation and selection were painless, and observation of the phenotype of the mutant obtained was extremely gratifying. Pure joy seeing those huge bubbles with plenty of little cells (some of which have completely lost their OM attachment) roaming inside. Moments like these are the reason why we still love doing scientific research even though 90% of what we do fails.

This is probably my favourite CryoEM image of the bubble mutant. I still regret it didn’t make it to the submitted version of the paper… But well, if you have tomograms acquired on Titan Krioss, you won’t show flat images acquired on a mere Tecnai 20…
Of course, this wasn’t the end of the challenge. Next step: to construct a complementation vector. If the successful ones we present in the study are bearing the numbers pJW34 and pJW35, it means that there were also the pJW-twenties and the pJW-tens constructed (or not) before. Their common point: none of them worked. Months and months of work for nothing, which left plenty of unanswered questions like “why modifying two base pairs between the Shine-Dalgarno sequence and the start codon would completely abolish protein production?”. And just when I obtained my complementation vectors, I realised that I had no way of putting them inside my “bubble mutant” because it had completely lost its natural competence. This was one of these moments in life where one is convinced that “PhD” stands for “Pound Head on Desk”. Thankfully I was able to develop a protocol for conjugative transfer from Escherichia coli to V. parvula. But don’t be fooled: as the bubble mutant is so fragile, sometimes all the exconjugants would die before I could save them as a glycerol stock. And even saving them as glycerol stock provided no warranty – sometimes they would die anyway in the -80°C freezer. Relaunching cultures from glycerol stock and handling the fragile bubble mutant was also far from easy. I’m unable to count the number of times where I wanted to estimate the MIC or do growth curves, but one of the bubbly strains decided either not to regrow from the glycerol stock or to die during the resuspension in the liquid medium. Oh, and have I already mentioned that V. parvula is able to sometimes recombine its DNA in a way that the only thing you can do while looking at the resulting strain is bugging your eyes out while reciting “today’s show is brought to you by the letters W, T and F” (for a better effect, imagine Big Bird running in panic mode in the background)?
One part where, for a change, I didn’t encounter technical issues was the CryoEM. First attempt – first success, and so on. And the success story repeated each time I would bring my samples to Anna Sartori-Rupp. Over these years I started to love our CryoEM sessions: a day when you know that everything you do will work is a nice change of perspective, especially when spent in such nice company, in a scientific routine marked by endless failures.

With Anna Sartori-Rupp, we had some heated debates whether the ruptured vesicles with discontinuous membranes we saw sometimes in CryoEM were a biological reality or an artefact of blotting. This little experiment settled the debate once and for all. You’re looking at an epifluorescence image (red – FM 4-64 coloration of biological membranes, blue – DAPI coloration of DNA) of Veillonella parvula ΔompM1-3 mutant deposited on a R 2/2 grid, on the left before the blotting, and on the right after it. As you can see, after the blotting the big vesicles are gone (exploded like soap bubbles hitting the ground) and fragments of the outer membrane have adhered to the mesh grid (visible as the diffuse red fluorescence with the black spots in the place of mesh grid holes)
For the paper we estimated that standard fluorescence microscopy was not enough, as Veillonella is so small… Thus, we decided to bring on board the specialist of the fluorescent microscopy on anabolic steroids (professionally known as 3D-SIM), Nika Pende. As we realised that on top of being a light microscopy expert, she is also quite an artist, we trusted her with all our schematic figures too.

If you mess up the parameters while generating the final rendering of your 3D CryoET reconstruction, you can end up with some abstract art instead of a figure for a paper.
As the experiments were progressing very slowly, coupled with the time the Institut Pasteur had to shut because of the pandemics, I found finally the time to initiate myself to bioinformatics, with the precious help of the in silico people of the lab. When I started the project, I was a complete greenhorn, whose greatest “accomplishment” in bioinformatics was using BLAST and MEGA. When I ended it, I was fluent in Perl and Python, and all the command line phylogenetic tools. This allowed me to perform the initial analysis of the main OM attachment systems in Bacteria, which was then completed on a 10x bigger database containing all the uncultured diversity of candidate phyla by Najwa Taib, our bioinformatic expert. These analyses show a very interesting result: whilst Gracilicutes (including well-studied classical diderms such as E. coli) have evolved multiple redundant systems to tether their OM, Terrabacteria (which Veillonella and other less studied diderms belong to) rely only on OmpM. This might explain why all the diderm to monoderm transitions have happened exclusively in this clade. My bubble mutant provides a nice support to this scenario, as it might be on its way to completely lose its OM, opening the way to the craziest experiment ever attempted: to recreate the diderm-to-monoderm transition in the lab.
Our work opens many questions and research avenues:
Why did Gracilicutes evolve multiple redundant systems to attach their OM? Do they rely on their OM more than diderm Terrabacteria? Did this confer an advantage allowing them to diversify widely?
How did the last bacterial common ancestor (LBCA) attach its OM? Is it possible that OmpM represents the most ancient -and simple- mechanism?
Many of the ancestral systems for OM biogenesis may be still present in Veillonella and can now be studied in depth. Other may still remain to be discovered. Combined with large-scale bioinformatic analyses, this will also shed light on the origin and evolution of main processes involved in OM stability in various bacteria including pathogenic ones.
Finally, our work opens new perspectives to understand an even deeper transition in the history of prokaryotic cell envelopes, that between Bacteria and Archaea, and the (diderm? monoderm? other?) nature of the last universal common ancestor (LUCA).
Every time the circle of knowledge is enlarged by new discoveries, the interface between the known and unknown, that corresponds to the open scientific questions, grows too… More science is what we need!
References
- Bladen, H. A. & Mergenhagen, S. E. ULTRASTRUCTURE OF VEILLONELLA AND MORPHOLOGICAL CORRELATION OF AN OUTER MEMBRANE WITH PARTICLES ASSOCIATED WITH ENDOTOXIC ACTIVITY. J. Bacteriol. 88, 1482–1492 (1964).
- Knapp, S. et al. Natural Competence Is Common among Clinical Isolates of Veillonella parvula and Is Useful for Genetic Manipulation of This Key Member of the Oral Microbiome. Front. Cell. Infect. Microbiol. 7, 139 (2017).
- Taib, N. et al. Genome-wide analysis of the Firmicutes illuminates the diderm/monoderm transition. Nat. Ecol. Evol. 4, 1661–1672 (2020).




Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in