When I started my PhD project in 2013, I realised the importance of the One Health approach, the interconnection between humans, animals, plants and their shared environment. Moreover, it is known that animals are important reservoirs and potential disseminators of bacteria and their antimicrobial resistance and virulence genes. The idea to study the One Health system was fascinating. For that reason, I decided to study the relationship of those ecological niches through the genus Pseudomonas.
We collected faecal samples from a wide variety of healthy animals: deer, wild boars, pets, micromammals, farm animals… We isolated many Pseudomonas species, some of them belonging to non-clinical areas, except the pathogen Pseudomonas aeruginosa. The antimicrobial resistance in these strains was studied and, unlike the antimicrobial resistance data in clinical strains, our Pseudomonas strains showed low resistance levels to antibiotics. However, it was interesting to discover empty integron structures, giving us to think that, under antibiotic pressure, these mobile genetic elements are ready to take antimicrobial resistance genes and favour their spread between bacteria from different environments. For that reason, we can’t relax about the use of antibiotics.
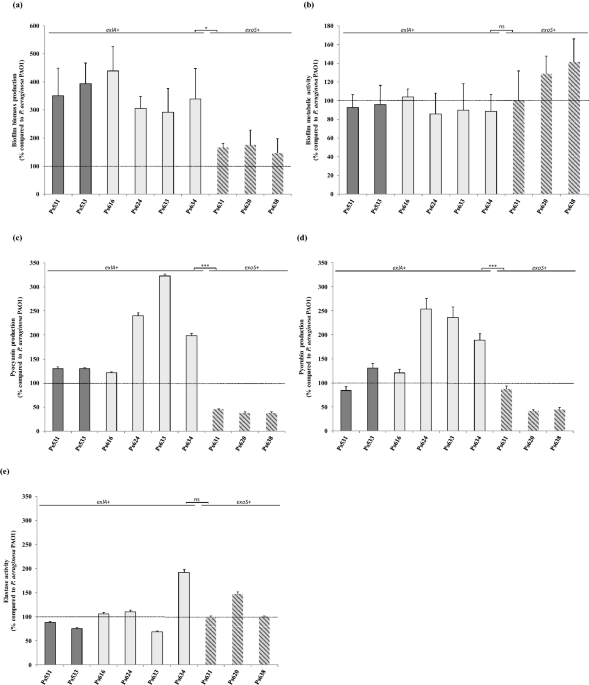
But, not always the problem is the antimicrobial resistance. What about the virulence? Pseudomonas possesses important virulence mechanisms, specially the species P. aeruginosa. In our work, regarding the selected nine P. aeruginosa strains, only three of them showed the Type 3 Secretion System (T3SS). This mechanism acts as a syringe delivering its toxins, termed effectors (ExoU, ExoS, ExoY and ExoT) directly into the cytoplasm of infected host cells. But, what happen with the remaining P. aeruginosa? Those strains harboured a new two partner secretion system (TP), called exolysin ExlA. This toxin, together with its cognate porin (ExlB), exhibits high cytotoxicity activity into host cells. To assess the pathogenic importance of these two systems, T3SS and TP, a cytotoxicity assay in two cell lines was carried out. Surprisingly, both cell lines were highly sensitive to most of the ExlA-positive strains. However, it is important to highlight that toxicity is strain‑dependent, and other factors could be involved, even other virulence mechanisms. For that reason, we had the idea to study the biofilm and pigments production, the elastase activity and their motility. The results showed that ExlA-positive strains exhibited higher virulence than T3SS-positive strains, but all of them described important virulence data.
A take-home message: although the environmental P. aeruginosa strains seem to be more antimicrobial susceptible and peaceful than the clinical ones, you can’t let your guard down. Bacteria can share these antimicrobial and virulence mechanisms with other pathogenic ones, as well as spread them into different environments. It is for this reason that the One Health approach was created.





Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in